Metallurgy: What’s the Difference Between Iron and Steel?
This article will discuss differences and similarities between wrought iron and steel. Although steel contains iron, it would be incorrect to merely view steel as a type of iron. The two are similar, but are not the same. These differences necessitate treating the two as distinct materials. This is important to note because there are few, if any, modern wrought iron producers in operation today. Any product on the market at this time would be a type of steel and would thus differ from a medieval shirt of maille which would have been made of wrought iron. This article will also discuss the implications of using low carbon steel rather than wrought iron on maille reproductions
Wrought Iron:
Wrought iron is a two component metal that contains high purity iron and iron silicate. [i] Although often merely called iron, wrought iron is actually a finished product containing ferrite, small amounts of carbon, and other elements. Iron, that is to say pure iron, does not exist in any commercial setting because it is too costly to truly remove all impurities from the iron. [ii] Another reason is that the impurities in wrought are actually beneficial. That’s where the iron silicate comes in. Iron silicate is itself an impurity in iron, which is a “glass-like slag.” [iii]
Unlike an alloy, where the two metals fuse on a chemical level, the combination of iron and iron silicate is only a physical association. That is to say, the two do not combine to form iron, but the two coexist in what we call iron. When wrought iron is produced, the iron in its heated state is “always composed of a pasty or semi-fused mass of cohering, slag-coated granules of refined iron.” [iv]
Iron silicate is a slag, but don’t think of slag as necessarily undesirable. Rather, the slag content, when properly controlled, is what makes iron ductile and workable. [v] The reason that the iron silicate slag remains in iron is that the furnaces in which the iron is produced do not liquefy the iron. As other impurities burn off, the iron silicate slag itself remains because neither the iron nor the slag reach a liquid state. The remaining slag helps to prevent an increased absorption of carbon. The increased absorption of carbon would in turn create a much stronger but less ductile metal.
Wrought iron typically contains less than .12% carbon but does retain 1-2% of iron silicate slag as well as small amounts of manganese, sulfur, and phosphorous. [vi] It also typically contains less carbon than steel, although low carbon steel does come close to the same content. [vii] Another feature of wrought iron is that it is not amenable to heat treatment. [viii] Thus, true tempering and can only be done with steel. Wrought iron will still maintain the same ductility whether quenched or not. However, that is not to say that wrought iron cannot be heat treated after work hardened to return its microstructure to a normal state.
Steel:
On the other hand, steel contains about 98% or more of iron and has very small levels of slag or impurities. [ix] Its carbon levels range from 1.5% to roughly 0.2%. [x] Another characteristic of steel is that steel can be worked directly or indirectly. That is to say, steel can be forged at a red heat – directly; or steel can be liquefied, casted, and molded – indirectly. In contrast to wrought iron, steel is extremely amenable to heat treatment. This is because steel contains higher carbon content than iron. [xi]
Perhaps most important, steel is an alloy. Wrought iron has both pure iron and iron silicate as distinct elements in wrought iron. On the other hand, steel is made through the chemical bonding of iron with carbon. This makes steel an alloy in the same way the bronze is an alloy of copper and tin. I point this out because it is not readily observable because the charcoal serves as both the heat source and the alloying material. [xii] Indeed, it was not until the 18th century that the role of carbon in steel was even widely accepted. [xiii]
Carbon: With all the mention of carbon content in iron and steel, we would do well to discuss carbon and what exactly it does to iron and steel. The higher the carbon content in either, the stronger the metal becomes. [xiv]. Steel has a higher carbon content than iron, but if the carbon content gets too high, above 2.2%, it will fall into the classification of cast iron. [xv] That is to say, a type of metal so carbon rich that it can only be worked through liquefaction and cast forming.
Those who have dealt with cast iron know that it is extremely hard, but that hardness has a drawback in reducing the malleability and ductility of the metal. [xvi] Therefore, although steel is stronger than iron, steel is also less flexible and malleable than iron. This makes steel generally harder to work with but gives it greater strength for uses in structural applications.
Steel can be made with varying carbon content, which gives it different properties that are useful for different applications. The carbon content difference between a mild steel and a high carbon steel is only about 0.35%. [xvii] Low carbon steel has less than 0.25% carbon while high carbon steel has more than 0.60% carbon. [xviii]
Wrought Iron:
Wrought iron is a two component metal that contains high purity iron and iron silicate. [i] Although often merely called iron, wrought iron is actually a finished product containing ferrite, small amounts of carbon, and other elements. Iron, that is to say pure iron, does not exist in any commercial setting because it is too costly to truly remove all impurities from the iron. [ii] Another reason is that the impurities in wrought are actually beneficial. That’s where the iron silicate comes in. Iron silicate is itself an impurity in iron, which is a “glass-like slag.” [iii]
Unlike an alloy, where the two metals fuse on a chemical level, the combination of iron and iron silicate is only a physical association. That is to say, the two do not combine to form iron, but the two coexist in what we call iron. When wrought iron is produced, the iron in its heated state is “always composed of a pasty or semi-fused mass of cohering, slag-coated granules of refined iron.” [iv]
Iron silicate is a slag, but don’t think of slag as necessarily undesirable. Rather, the slag content, when properly controlled, is what makes iron ductile and workable. [v] The reason that the iron silicate slag remains in iron is that the furnaces in which the iron is produced do not liquefy the iron. As other impurities burn off, the iron silicate slag itself remains because neither the iron nor the slag reach a liquid state. The remaining slag helps to prevent an increased absorption of carbon. The increased absorption of carbon would in turn create a much stronger but less ductile metal.
Wrought iron typically contains less than .12% carbon but does retain 1-2% of iron silicate slag as well as small amounts of manganese, sulfur, and phosphorous. [vi] It also typically contains less carbon than steel, although low carbon steel does come close to the same content. [vii] Another feature of wrought iron is that it is not amenable to heat treatment. [viii] Thus, true tempering and can only be done with steel. Wrought iron will still maintain the same ductility whether quenched or not. However, that is not to say that wrought iron cannot be heat treated after work hardened to return its microstructure to a normal state.
Steel:
On the other hand, steel contains about 98% or more of iron and has very small levels of slag or impurities. [ix] Its carbon levels range from 1.5% to roughly 0.2%. [x] Another characteristic of steel is that steel can be worked directly or indirectly. That is to say, steel can be forged at a red heat – directly; or steel can be liquefied, casted, and molded – indirectly. In contrast to wrought iron, steel is extremely amenable to heat treatment. This is because steel contains higher carbon content than iron. [xi]
Perhaps most important, steel is an alloy. Wrought iron has both pure iron and iron silicate as distinct elements in wrought iron. On the other hand, steel is made through the chemical bonding of iron with carbon. This makes steel an alloy in the same way the bronze is an alloy of copper and tin. I point this out because it is not readily observable because the charcoal serves as both the heat source and the alloying material. [xii] Indeed, it was not until the 18th century that the role of carbon in steel was even widely accepted. [xiii]
Carbon: With all the mention of carbon content in iron and steel, we would do well to discuss carbon and what exactly it does to iron and steel. The higher the carbon content in either, the stronger the metal becomes. [xiv]. Steel has a higher carbon content than iron, but if the carbon content gets too high, above 2.2%, it will fall into the classification of cast iron. [xv] That is to say, a type of metal so carbon rich that it can only be worked through liquefaction and cast forming.
Those who have dealt with cast iron know that it is extremely hard, but that hardness has a drawback in reducing the malleability and ductility of the metal. [xvi] Therefore, although steel is stronger than iron, steel is also less flexible and malleable than iron. This makes steel generally harder to work with but gives it greater strength for uses in structural applications.
Steel can be made with varying carbon content, which gives it different properties that are useful for different applications. The carbon content difference between a mild steel and a high carbon steel is only about 0.35%. [xvii] Low carbon steel has less than 0.25% carbon while high carbon steel has more than 0.60% carbon. [xviii]
 Sauveur, Metallography and Heat Treatment, 54.
Sauveur, Metallography and Heat Treatment, 54.
Microstructure:
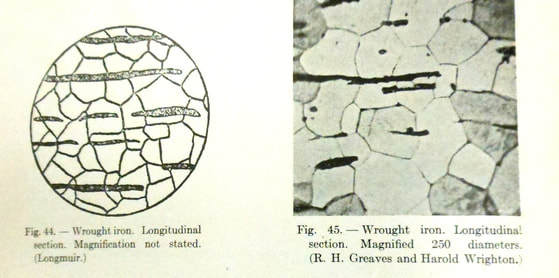
What then is the difference between low carbon steel and wrought iron? To explain this, one must look at the microstructure of each metal. Cross section magnification of wrought iron shows irregular black lines, “varying much in thickness and length, but all running in the same direction * * * These lines indicate the location of the slag which has assumed the shape of fibers or streaks running in the direction of the rolling or forging, thus imparting a fibrous appearance to the metal.” [i] The carbon in wrought iron is difficult t distinguish from the slag. [ii]
What then is the difference between low carbon steel and wrought iron? To explain this, one must look at the microstructure of each metal. Cross section magnification of wrought iron shows irregular black lines, “varying much in thickness and length, but all running in the same direction * * * These lines indicate the location of the slag which has assumed the shape of fibers or streaks running in the direction of the rolling or forging, thus imparting a fibrous appearance to the metal.” [i] The carbon in wrought iron is difficult t distinguish from the slag. [ii]
 Sauveur, Metallography and Heat Treatment, 56.
Sauveur, Metallography and Heat Treatment, 56.
By examining the microstructure of carbon you can see the wood-grain appearance in the metal. The three dimensional cross section shows that the slag inclusions run the length of the wrought iron.
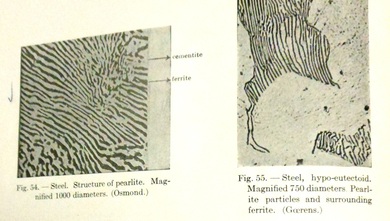
A microstructure examination of low carbon steel reveals a different picture, although steel’s microstructure contains dark areas, these areas are not actually free carbon. Remember that the carbon in steel has combined with the iron. Rather, the dark areas in the microstructure of steel are the carbon and iron alloyed. [i] The structure is called pearlite due to its pearly appearance when examined. The contrast of the light colored carbon (also called cementite) and the dark colored ferrite actually gives an alternating and pearly glisten when viewed in certain lights under magnification. [ii]
A microstructure examination of low carbon steel reveals a different picture, although steel’s microstructure contains dark areas, these areas are not actually free carbon. Remember that the carbon in steel has combined with the iron. Rather, the dark areas in the microstructure of steel are the carbon and iron alloyed. [i] The structure is called pearlite due to its pearly appearance when examined. The contrast of the light colored carbon (also called cementite) and the dark colored ferrite actually gives an alternating and pearly glisten when viewed in certain lights under magnification. [ii]
Pearlite in steel increases proportionally to the amount of carbon in steel. Thus the more carbon in steel, the more
apparent pearlite will be. [i] The inverse of this is that the more pearlite in steel, the less iron the steel will contain.
Upon microstructure examination of wrought iron and a comparison of microstructure examination with steel, the conclusion is that the ferrite structure – that is to say the iron itself – “is in no way different from the ferrite forming the bulk of low carbon steel. Both are equally crystalline.” [ii] However, wrought iron contains the wood-like slag inclusions while steel contains the pearlite structure of carbon and ferrite alloyed.
Remember that the slag inclusions in wrought iron appear as wood grains in the structure of the metal. These slag inclusions give wrought iron increased ductility which allows the iron to bend but not break. Wrought iron will bend like dough rather than snap like a stick. [iii] Moreover, the slag inclusions also help to prevent corrosion in wrought iron. This improves the longevity of wrought iron over that of steel, which does not include the slag. [iv]
One might wonder why a low carbon steel would not similarly bend or have a dough-like structure as wrought iron. The reason again comes back to steel being an alloy. Although the total carbon content might be nearly the same, the fact that wrought iron has slag inclusions means that the carbon content of .02% in wrought iron is mostly contained in the slag itself rather than the iron. This means that the ferrite in wrought iron is purer than the ferrite in steel. The carbon in steel, as shown in the pearlite structure of steel, is spread evenly, which increases the overall strength of steel but decreases its overall ductility – even if the total carbon content is exactly the same for a given sample of wrought iron and steel.
apparent pearlite will be. [i] The inverse of this is that the more pearlite in steel, the less iron the steel will contain.
Upon microstructure examination of wrought iron and a comparison of microstructure examination with steel, the conclusion is that the ferrite structure – that is to say the iron itself – “is in no way different from the ferrite forming the bulk of low carbon steel. Both are equally crystalline.” [ii] However, wrought iron contains the wood-like slag inclusions while steel contains the pearlite structure of carbon and ferrite alloyed.
Remember that the slag inclusions in wrought iron appear as wood grains in the structure of the metal. These slag inclusions give wrought iron increased ductility which allows the iron to bend but not break. Wrought iron will bend like dough rather than snap like a stick. [iii] Moreover, the slag inclusions also help to prevent corrosion in wrought iron. This improves the longevity of wrought iron over that of steel, which does not include the slag. [iv]
One might wonder why a low carbon steel would not similarly bend or have a dough-like structure as wrought iron. The reason again comes back to steel being an alloy. Although the total carbon content might be nearly the same, the fact that wrought iron has slag inclusions means that the carbon content of .02% in wrought iron is mostly contained in the slag itself rather than the iron. This means that the ferrite in wrought iron is purer than the ferrite in steel. The carbon in steel, as shown in the pearlite structure of steel, is spread evenly, which increases the overall strength of steel but decreases its overall ductility – even if the total carbon content is exactly the same for a given sample of wrought iron and steel.
An alloy has properties of both of its constituents. Ferrite in its pure form has a Brinell hardness of about 80. [1] Carbon, by contrast, has a hardness of 700 or more. [2] Meanwhile, pearlite, the alloying of carbon and ferrite, has a Brinell hardness of 200. This means that steel is much harder that wrought iron, but this hardness comes at the price of ductility, which is measured by only 10% elongation per 2 inches in steel versus 50% elongation per two inches in ferrite. [3] Another point to mention is that this applies only to steel in its pearlite form – that is to say, steel that has been “worked, reheated to a high temperature, and slowly cooled.” [4] Steel that has been cold worked, quenched, or tempered, will not retain the same properties but can typically be returned through annealing.
At bottom then, there are three key differences between wrought iron and low carbon steel. First, wrought iron contains slag impurities that are not burned off while steel does not contain these impurities. The slag in wrought iron allows for the iron to be in a more pure state which increases wrought iron’s ductility but reduces its hardness. This leads to the second difference, which is that wrought iron is never produced through liquefaction. In fact, it cannot be because to liquefy it would remove its hallmark feature, the iron silicate. Finally, wrought iron will not change structure due to heat treatment while steel is amenable to heat treatment.
Effect on Maille:
Finally, there is the question of what effect, if any, this has on historical maille made from wrought iron versus maille made today with low carbon steel. First, maille made from wrought iron would likely rust at a lower rate than maille made from modern steel. This is because wrought iron has higher concentrations of pure iron which rusts more slowly than steel. The effect of this would be that modern maille will likely not be found 500 years from now in any salvageable sate.
Second, the increased ductility would allow maille to absorb more of a blow before cracking or snapping apart. The dough-like nature of wrought iron would allow the links to bend but not break apart. This might help to prevent holes during battle and would also likely absorb more of the energy of the blow rather than transferring it directly to the wearer. This is similar to the difference between a modern car bumper and an old style steel bumper. In old fashioned car bumpers, the bumper has no give whatsoever, the impact will reduce damage to the car but will actually increase injury to the occupants because none of the impact was absorbed. Conversely, in the modern car, the bumper is designed to crumple upon impact. The reason for this is that the crumpling of the bumper will help to absorb the damaging effects of the impact.
At bottom then, there are three key differences between wrought iron and low carbon steel. First, wrought iron contains slag impurities that are not burned off while steel does not contain these impurities. The slag in wrought iron allows for the iron to be in a more pure state which increases wrought iron’s ductility but reduces its hardness. This leads to the second difference, which is that wrought iron is never produced through liquefaction. In fact, it cannot be because to liquefy it would remove its hallmark feature, the iron silicate. Finally, wrought iron will not change structure due to heat treatment while steel is amenable to heat treatment.
Effect on Maille:
Finally, there is the question of what effect, if any, this has on historical maille made from wrought iron versus maille made today with low carbon steel. First, maille made from wrought iron would likely rust at a lower rate than maille made from modern steel. This is because wrought iron has higher concentrations of pure iron which rusts more slowly than steel. The effect of this would be that modern maille will likely not be found 500 years from now in any salvageable sate.
Second, the increased ductility would allow maille to absorb more of a blow before cracking or snapping apart. The dough-like nature of wrought iron would allow the links to bend but not break apart. This might help to prevent holes during battle and would also likely absorb more of the energy of the blow rather than transferring it directly to the wearer. This is similar to the difference between a modern car bumper and an old style steel bumper. In old fashioned car bumpers, the bumper has no give whatsoever, the impact will reduce damage to the car but will actually increase injury to the occupants because none of the impact was absorbed. Conversely, in the modern car, the bumper is designed to crumple upon impact. The reason for this is that the crumpling of the bumper will help to absorb the damaging effects of the impact.
[1] James Aston, Wrought Iron: Its Manufacture, Characteristics and Applications, (Pittsburgh: A.M. Byers Co., 1957): 1.
[2] G. Reginald Bashforth, The Manufacture of Iron and Steel, (London: Chapman & Hall, 1948), 4.
[3] Aston, Wrought Iron, 1.
[4] Ibid.
[5] Ibid. at 2.
[6] Ibid.
[7] Ibid.
[8] G. Reginald Bashforth, The Manufacture of Iron and Steel, (London: Chapman & Hall, 1948), 4.
[9] Ibid.
[10] Ibid.
[11] Albert Sauveur, The Metallography and Heat Treatment of Iron and Steel, (New York: McGraw Hill, 1935): 54.
[12] Cyril Stanley Smith, “The discovery of Carbon in Steel,” Technology and Culture, Vol. 5 No. 2, Johns Hopkins Univ. Press, (Johns Hopkins, 1964): 150.
[13] Ibid.
[14] Bashforth, Manufacture of Iron and Steel, 6.
[15] Ibid.
[16] Ibid.
[17] Sauveur, Metallography and Heat Treatment, 59.
[18] Ibid.
[19] Ibid. at 55.
[20] Ibid.
[21] Ibid. at 61.
[22] Ibid. at 62.
[23] Ibid. at 66.
[24] Ibid. at 56.
[25] Todd Daniel, “Clearing the Confusion Over Wrought Iron,” Fabricator, http://www.artmetal.com/project/NOMMA/WROUGHT.HTM, (Accessed 6/16/14).
[26] Ibid.
[27] Sauveur, Metallography and Heat Treatment , 79.
[28] Ibid.
[29] Ibid.
[30] Ibid. at 80.
[2] G. Reginald Bashforth, The Manufacture of Iron and Steel, (London: Chapman & Hall, 1948), 4.
[3] Aston, Wrought Iron, 1.
[4] Ibid.
[5] Ibid. at 2.
[6] Ibid.
[7] Ibid.
[8] G. Reginald Bashforth, The Manufacture of Iron and Steel, (London: Chapman & Hall, 1948), 4.
[9] Ibid.
[10] Ibid.
[11] Albert Sauveur, The Metallography and Heat Treatment of Iron and Steel, (New York: McGraw Hill, 1935): 54.
[12] Cyril Stanley Smith, “The discovery of Carbon in Steel,” Technology and Culture, Vol. 5 No. 2, Johns Hopkins Univ. Press, (Johns Hopkins, 1964): 150.
[13] Ibid.
[14] Bashforth, Manufacture of Iron and Steel, 6.
[15] Ibid.
[16] Ibid.
[17] Sauveur, Metallography and Heat Treatment, 59.
[18] Ibid.
[19] Ibid. at 55.
[20] Ibid.
[21] Ibid. at 61.
[22] Ibid. at 62.
[23] Ibid. at 66.
[24] Ibid. at 56.
[25] Todd Daniel, “Clearing the Confusion Over Wrought Iron,” Fabricator, http://www.artmetal.com/project/NOMMA/WROUGHT.HTM, (Accessed 6/16/14).
[26] Ibid.
[27] Sauveur, Metallography and Heat Treatment , 79.
[28] Ibid.
[29] Ibid.
[30] Ibid. at 80.